Arenes
Arenes (also known as aromatic hydrocarbons) are hydrocarbons that contain in their molecule one or more rings with delocalized pi electrons that constitute the aromaticity. An example is the ring with 6 carbon atoms (this being called the benzene nucleus). The name aromatics predates the knowledge of the electronic structure of aromatic cycles.
When the arene molecule consists of a single ring, they are called mononuclear arenes, and when the molecule contains two or more rings, they are called polynuclear or polycyclic arenes. When the atoms of other elements appear in the aromatic rings, they are heterocyclic aromatic compounds, such as pyridine, pyrimidine, etc.
Classification
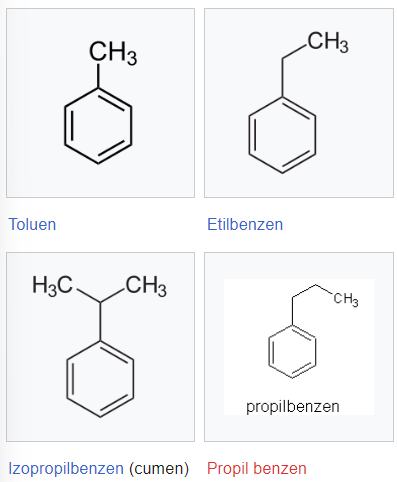
Mononuclear arenes
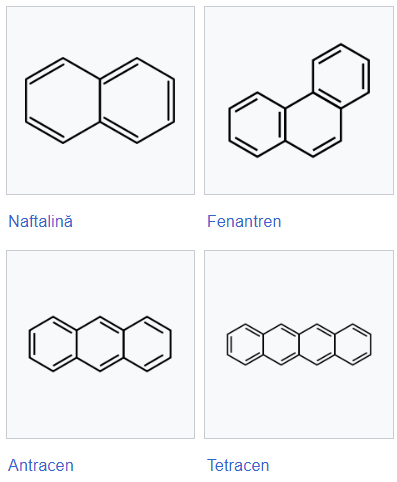
Polynuclear arenes
Structure and nomenclature
For mononuclear aromatic hydrocarbons, the head of the series is benzene C6H6, the higher homologue being methylbenzene (toluene), followed by ethylbenzene, n-propylbenzene, isopropylbenzene (cumene) in the homologous series. For substituted derivatives of benzene there are 3 types of isomers: ortho (1,2) or orthoxylene, meta (1,3) or metaxylene para (1,4) paraxylene.
Synthesis of arenes
Pyrolysis
The pyrolysis of organic substances is one of the most important ways of obtaining arenes with wood as raw material, but more so coal. Dry distillation or coal calcination includes complex processes that take place at temperatures of 500-1200 °C, under vacuum, obtaining the following fractions:
According to the temperature at which this pyrolysis process takes place, they can be distinguished:
Various processes result in an absorption oil saturated with hydrocarbons; this is heated to 135-140 °C and then distilled with superheated steam in a distillation column. The distillation product up to 140 °C – consisting of benzene, toluene, xylene – represents crude benzene. The mixture with boiling points above 140 °C (naphthalene, aromatic compounds with sulfur nitrogen oxygen) forms a good solvent mixture known as solvent naphtha.
Coal tars
They are another important source of raw material for the chemical industry; they appear as an oily, black-brown, very viscous liquid, with a characteristic, unpleasant and penetrating smell. Tars are made up of numerous organic substances that can be grouped as follows:
Along with these components, there is also free carbon in the form of carbon black, the higher this percentage of free carbon, the lower the quality of the tar. Tar processing can be done by:
Oil distillation
It is another source of production, due to the fact that the percentage of benzene and xylenes provided by the 2 previous methods is no longer sufficient. Due to the fact that the percentage of arenes in crude oil is small, this type of hydrocarbon is subjected to a reforming process ( cracking), in the presence of a catalyst, usually platinum, hence the name of the platforming process (or platforming). The reactions that occur during the process are cyclization, followed by dehydrogenation, both catalyzed by platinum. Cycloalkanes with a 6-carbon ring undergo direct cyclization. The catalytic reforming process is followed by the rather cumbersome separation at the beginning of the history of this method. A separation process was developed by the Romanian engineer Lazăr Edeleanu (the Edeleanu process), in which SO2 is used as a separation medium that at low temperatures dissolves only aromatic hydrocarbons. Glycol is currently preferred as the separation medium.
Catalytic dehydrogenation
It is one of the most modern methods of obtaining aromatic hydrocarbons, also known as catalytic reforming. As a result of the process, reformed gasolines are obtained consisting of a mixture of hydrocarbons, being used for engines (having a high octane number) or for obtaining aromatic hydrocarbons.